High-temperature decomposition of Cu2BaSnS4 with Sn loss reveals newly identified compound Cu2Ba3Sn2S8†
Abstract
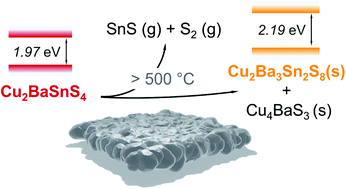
The earth-abundant quaternary compound Cu2BaSnS4 is being currently studied as a candidate for photovoltaics and as a photocathode for water splitting. However, the chemical stability of this phase during synthesis is unclear. The synthesis of other quaternary tin–sulphur-based absorbers (e.g., Cu2ZnSnS4) involves an annealing step at high temperature under sulphur gas atmosphere, which can lead to decomposition into secondary phases involving Sn loss from the sample. As the presence of secondary phases can be detrimental for device performance, it is crucial to identify secondary phase chemical, structural and optoelectronic properties. Here we used a combination of in situ EDXRD/XRF and TEM to identify a decomposition pathway for Cu2BaSnS4. Our study reveals that, while Cu2BaSnS4 remains stable at high sulphur partial pressure, the material decomposes at high temperatures into Cu4BaS3 and the hitherto unknown compound Cu2Ba3Sn2S8 if the synthesis is performed under low partial pressure of sulphur. The presence of Cu4BaS3 in devices could be harmful due to its high conductivity and relatively lower band gap compared to Cu2BaSnS4. The analysis of powder diffraction data reveals that the newly identified compound Cu2Ba3Sn2S8 crystallizes in the cubic system (space group I![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) 3d) with a lattice parameter of a = 14.53(1) Å. A yellow powder of Cu2Ba3Sn2S8 has been synthesized, exhibiting an absorption onset at 2.19 eV.
3d) with a lattice parameter of a = 14.53(1) Å. A yellow powder of Cu2Ba3Sn2S8 has been synthesized, exhibiting an absorption onset at 2.19 eV.


 Please wait while we load your content...
Please wait while we load your content...