Degradation mechanisms in mixed-cation and mixed-halide CsxFA1−xPb(BryI1−y)3 perovskite films under ambient conditions†
Abstract
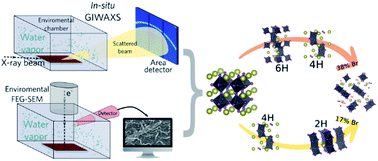
Multicomponent perovskites of the type CsxFA1−xPb(BryI1−y)3 are good candidates for highly efficient perovskite and tandem solar cells. In this work the degradation mechanisms of these multicomponent films were investigated and our results show that the degradation is a complex process, with the formation of a number of intermediates and lead-based products. In situ X-ray diffraction analysis carried out in the first stages of the degradation indicate that different from MAPbI3 perovskites, the degradation of these multicomponent films begins with the formation of hexagonal polytypes as intermediates, which in turn are converted to hydrated phases. The initial steps of the degradation were also monitored for the first time by in situ environmental scanning electron microscopy (ESEM) with 75% of relative humidity. In situ ESEM images show that the degradation has its beginning at the “valleys” of the wrinkled morphology found in these films, possibly because of a smaller grain size in these regions. XPS analysis confirms that the hydrated perovskite films continue to react with the environment, leading to the formation of metal hydroxides, carbonates, and oxides as final products. Our results also indicate that the degradation mechanism is highly dependent on the Cs concentration and Br content providing guidance for choosing the best compositions for efficient, but more environmentally stable solar cells.
- This article is part of the themed collections: Celebrating Latin American Talent in Chemistry and Journal of Materials Chemistry A Advisory Board Collection


 Please wait while we load your content...
Please wait while we load your content...