A "trimurti" heterostructured hybrid with an intimate CoO/CoxP interface as a robust bifunctional air electrode for rechargeable Zn–air batteries†
Abstract
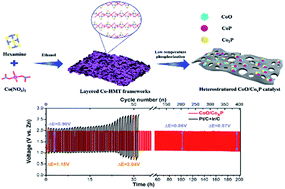
The development of robust and cost-effective bifunctional oxygen electrocatalysts is of significant importance for the widespread implementation of Zn–air batteries yet remains an immense challenge, which calls for the molecular-level manipulation of active species as well as morphology engineering to accelerate the reversible oxygen reaction dynamics. Herein, a “trimurti” heterostructured cobalt-based hybrid is designed by a facile, one-step phosphorization of layered Co-hexamine metal–organic frameworks. The synergistic effect between cobalt phosphides (Co2P and CoP) and cobalt oxide significantly boosts the electrocatalytic activity of the oxygen redox reaction. Besides, the hierarchically porous structure promotes the accessibility of active species and smooth electron/reactant transportation. Due to these attributes, the as-developed electrocatalyst outperforms the state-of-art non-noble metal catalysts and even noble metal benchmarks with a half-wave potential of 0.86 V for the ORR and an overpotential of 0.37 V at 10 mA cm−2 for the OER. Furthermore, an appealing catalytic performance is also demonstrated in an assembled Zn–air battery, which displays a lower voltage gap of 0.86 V and improved cyclability of 202 h. This work not only affords a competitive bifunctional oxygen electrocatalyst for Zn–air batteries but also highlights the synergetic effect from heterointerfaces in electrocatalysis.


 Please wait while we load your content...
Please wait while we load your content...