Doping mechanisms of N-DMBI-H for organic thermoelectrics: hydrogen removal vs. hydride transfer†
Abstract
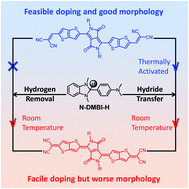
Efficient n-doping is a big challenge to organic thermoelectrics due to the requirements of both low ionization potentials and good air-stability. In recent years, N-DMBI-H has been reported as the most successful solution-processed n-type dopant precursor, but the doping mechanisms have been unclear to date. Here, we have investigated both hydrogen removal and hydride transfer reactions of N-DMBI-H doped in representative n-type molecular semiconductors (i.e., aromatic A-DCV-DPPTT and quinoid Q-DCM-DPPTT) by means of density functional theory calculations. The results demonstrate that N-DMBI-H is an air-stable n-type dopant precursor. Interestingly, when the N-DMBI-H precursor is associated with the semiconductor molecules, the reaction energy barriers for both hydrogen removal and hydride transfer are substantially decreased because of the assistance from the strong electron affinity of the semiconductors. In the case of the A-DCV-DPPTT/N-DMBI-H complexes, the hydride transfer reaction can be thermally activated. Notably, for the Q-DCM-DPPTT/N-DMBI-H complexes, both the chemical reactions can take place and bring about efficient charge transfer at room temperature. However, owing to the strong binding between the dopant and semiconductor molecules in the charge–transfer complexes, long-range ordered molecular packing will be disrupted for Q-DCM-DPPTT during solution processing, which is detrimental to charge transport; thus the experimental electrical conductivity is relatively lower for Q-DCM-DPPTT with respect to A-DCV-DPPTT. This work provides an in-depth understanding of the n-doping mechanisms of N-DMBI-H, paving the way towards rational development of both molecular dopants and semiconductors for high-performance organic thermoelectrics.


 Please wait while we load your content...
Please wait while we load your content...