The role of connectivity in significant bandgap narrowing for fused-pyrene based non-fullerene acceptors toward high-efficiency organic solar cells†
Abstract
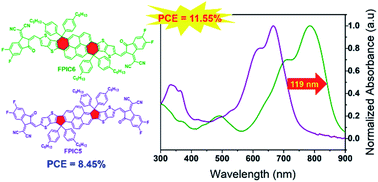
Great attention has been paid to developing low bandgap non-fullerene acceptors (NFAs) for matching wide bandgap donor polymers to increase the photocurrent and therefore the power conversion efficiencies (PCEs) of NFA organic solar cells, while pyrene-core based acceptor–donor–acceptor (A–D–A) NFAs have been mainly reported via the 2,9-position connection due to their bisthieno[3′,2′-b']thienyl[a,h]pyrene fused via a five-membered ring bridge at the ortho-position of pyrene as the representative one named FPIC5, which has prohibited further narrowing their energy gap. Herein, an acceptor FPIC6 was exploited by creating the 1,8-position connection through fusing as bisthieno[3′,2′-b′]thienyl[f-g,m-n]pyrene linked at the bay-position via a six-membered bridge, with enhanced push–pull characteristics within such A–D–A structure. As a structural isomer of FPIC5, FPIC6 exhibited a much lower bandgap of 1.42 eV (1.63 eV for FPIC5). Therefore, the photocurrent and PCE of PTB7-Th:FPIC6 cells were improved to 21.50 mA cm−2 and 11.55%, respectively, due to the balanced mobilities, better photoluminescence quenching efficiency and optimized morphology, which are both ∼40% better than those of PTB7-Th:FPIC5 cells. Our results clearly proved that a pyrene fused core with 1,8-position connection with electron-withdrawing end groups instead of 2,9-position connection is an efficient molecular design strategy to narrow the optical bandgap and improve the photovoltaic performance of NFA based OSCs.


 Please wait while we load your content...
Please wait while we load your content...