Engineering the hydrogen evolution reaction of transition metals: effect of Li ions†
Abstract
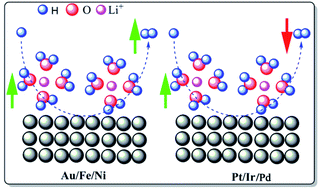
Though the role of supporting ions such as Li ions (Li+) in the electrochemical hydrogen evolution reaction (HER) has been extensively studied in the recent past, the fundamental mechanism influencing the HER kinetics of different metals under varied electrolyte conditions is still not known. Here we unveil the mechanism leading to a tunable HER on different metals under varying pH conditions, and it is established that despite the nominal role of counter anions, Li+ alone can tune both the HER thermodynamics and kinetics of metals. Different metals, namely Pt, Ir, Pd, Au, Fe, and Ni, are studied here to ensure the representation from both the sides of the Sabatier HER plot, under different pH conditions towards their HER efficacy at varying Li+ concentrations. Pt, Pd, and Ir showed suppression in their HER properties with increasing Li+ concentration, while the reverse phenomenon was observed on the rest of the metals. The metal–hydrogen (M–H) binding energy variation with Li+ is theoretically proven, and the Pt–H and Pd–H binding energy variations with Li+ containing electrolytes are experimentally demonstrated. Studies show that the tunability of the HER properties of both noble and non-noble metals can be achieved irrespective of the pH (0 and 13) and counter ions (TFSI−, Cl−, ClO4−, NO3− and OH−) by tuning the M–H bond energy using Li+, where this will be helpful in designing better electro-catalysts not only for the HER but also for other important reactions such as carbon dioxide reduction, nitrogen reduction, etc., where the HER is their competitor in aqueous medium.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...