Molecular-level insight in supported olefin metathesis catalysts by combining surface organometallic chemistry, high throughput experimentation, and data analysis†
Abstract
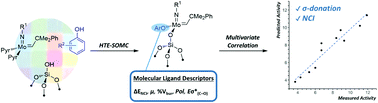
A combination of high-throughput experimentation (HTE), surface organometallic chemistry (SOMC) and statistical data analysis provided the platform to analyze in situ silica-grafted Mo imido alkylidene catalysts based on a library of 35 phenols. Overall, these tools allowed for the identification of σ-donor electronic effects and dispersive interactions and as key drivers in a prototypical metathesis reaction, homodimerization of 1-nonene. Univariate and multivariate correlation analysis confirmed the categorization of the catalytic data into two groups, depending on the presence of aryl groups in ortho position of the phenol ligand. The initial activity (TOFin) was predominantly correlated to the σ-donor ability of the aryloxy ligands, while the overall catalytic performance (TON1 h) was mainly dependent on attractive dispersive interactions with the used phenol ligands featuring aryl ortho substituents and, in sharp contrast, repulsive dispersive interactions with phenol free of aryl ortho substituents. This work outlines a fast and efficient workflow of gaining molecular-level insight into supported metathesis catalysts and highlights σ-donor ability and noncovalent interactions as crucial properties for designing active d0 supported metathesis catalysts.
- This article is part of the themed collection: 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...