Supramolecular catalysis by recognition-encoded oligomers: discovery of a synthetic imine polymerase†
Abstract
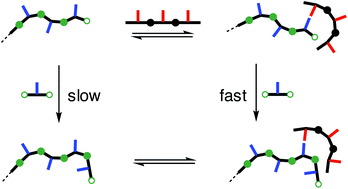
All key chemical transformations in biology are catalysed by linear oligomers. Catalytic properties could be programmed into synthetic oligomers in the same way as they are programmed into proteins, and an example of the discovery of emergent catalytic properties in a synthetic oligomer is reported. Dynamic combinatorial chemistry experiments designed to study the templating of a recognition-encoded oligomer by the complementary sequence have uncovered an unexpected imine polymerase activity. Libraries of equilibrating imines were formed by coupling diamine linkers with monomer building blocks composed of dialdehydes functionalised with either a trifluoromethyl phenol (D) or phosphine oxide (A) H-bond recognition unit. However, addition of the AAA trimer to a mixture of the phenol dialdehyde and the diamine linker did not template the formation of the DDD oligo-imine. Instead, AAA was found to be a catalyst, leading to rapid formation of long oligomers of D. AAA catalysed a number of different imine formation reactions, but a complementary phenol recognition group on the aldehyde reaction partner is an essential requirement. Competitive inhibition by an unreactive phenol confirmed the role of H-bonding in substrate recognition. AAA accelerates the rate of imine formation in toluene by a factor of 20. The kinetic parameters for this enzyme-like catalysis are estimated as 1 × 10−3 s−1 for kcat and the dissociation constant for substrate binding is 300 μM. The corresponding DDD trimer was found to catalyse oligomerisation the phosphine oxide dialdehyde with the diamine linker, suggesting an important role for the backbone in catalysis. This unexpected imine polymerase activity in a duplex-forming synthetic oligomer suggests that there are many interesting processes to be discovered in the chemistry of synthetic recognition-encoded oligomers that will parallel those found in natural biopolymers.


 Please wait while we load your content...
Please wait while we load your content...