Comparison of tetravalent cerium and terbium ions in a conserved, homoleptic imidophosphorane ligand field†
Abstract
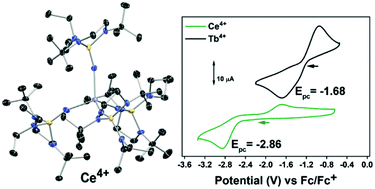
A redox pair of Ce4+ and Ce3+ complexes has been prepared that is stabilized by the [(NP(1,2-bis-tBu-diamidoethane)(NEt2))]1− ligand. Since these complexes are isostructural to the recently reported isovalent terbium analogs, a detailed structural and spectroscopic comparative analysis was pursued via Voronoi–Dirichlet polyhedra analysis, UV-vis-NIR, L3-edge X-ray absorption near edge spectroscopy (XANES), cyclic voltammetry, and natural transitions orbital (NTO) analysis and natural bond orbital (NBO) analysis. The electrochemical studies confirm previous theoretical studies of the redox properties of the related complex [K][Ce3+(NP(pip)3)4] (pip = piperidinyl), 1-Ce(PN). Complex 1-Ce(PN*) presents the most negative Epc of −2.88 V vs. Fc/Fc+ in THF of any cerium complex studied electrochemically. Likewise 1-Tb(PN*) has the most negative Epc for electrochemically interrogated terbium complexes at −1.79 V vs. Fc/Fc+ in THF. Complexes 1-Ce(PN*) and 2-Ce(PN*) were also studied by L3-edge X-ray absorption near edges spectroscopy (XANES) and a comparison to previously reported spectra for 1-Tb(PN*), 2-Tb(PN*), 1-Ce(PN), and, [Ce4+(NP(pip)3)4], 2-Ce(PN), demonstrates similar nf values for all the tetravalent lanthanide complexes. According to the natural bond orbital analysis, a greater covalent character of the M–L bonds is found in 2-Ce(PN*) than in 1-Ce(PN*), in agreement with the shorter Ce–N bonds in the tetravalent counterpart. The greater contribution of Ce orbitals in the Ce–N bonding and, specifically, the higher participation of 4f electrons accounts for the stronger covalent interactions in 2-Ce(PN*) as compared to 2-Tb(PN*).
- This article is part of the themed collection: 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...