A soft molecular 2Fe–2As precursor approach to the synthesis of nanostructured FeAs for efficient electrocatalytic water oxidation†
Abstract
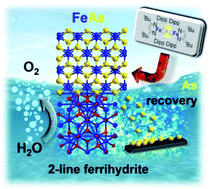
An unprecedented molecular 2Fe–2As precursor complex was synthesized and transformed under soft reaction conditions to produce an active and long-term stable nanocrystalline FeAs material for electrocatalytic water oxidation in alkaline media. The 2Fe2As-centred β-diketiminato complex, having an unusual planar Fe2As2 core structure, results from the salt-metathesis reaction of the corresponding β-diketiminato FeIICl complex and the AsCO− (arsaethynolate) anion as the monoanionic As− source. The as-prepared FeAs phase produced from the precursor has been electrophoretically deposited on conductive electrode substrates and shown to act as a electro(pre)catalyst for the oxygen evolution reaction (OER). The deposited FeAs undergoes corrosion under the severe anodic alkaline conditions which causes extensive dissolution of As into the electrolyte forming finally an active two-line ferrihydrite phase (Fe2O3(H2O)x). Importantly, the dissolved As in the electrolyte can be fully recaptured (electro-deposited) at the counter electrode making the complete process eco-conscious. The results represent a new and facile entry to unexplored nanostructured transition-metal arsenides and their utilization for high-performance OER electrocatalysis, which are also known to be magnificent high-temperature superconductors.


 Please wait while we load your content...
Please wait while we load your content...