Copper-mediated peptide arylation selective for the N-terminus†
Abstract
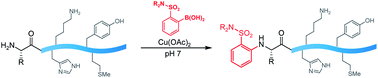
Polypeptides present remarkable selectivity challenges for chemical methods. Amino groups are ubiquitous in polypeptide structure, yet few paradigms exist for reactivity and selectivity in arylation of amine groups. This communication describes the utilization of boronic acid reagents bearing certain o-electron withdrawing groups for copper-mediated amine arylation of the N-terminus under mild conditions and primarily aqueous solvent. The method adds to the toolkit of boronic acid reagents for polypeptide modification under mild conditions in water that shows complete selectivity for the N-terminus in the presence of lysine side chains.


 Please wait while we load your content...
Please wait while we load your content...