Pnictogen-bonding catalysis: brevetoxin-type polyether cyclizations†
Abstract
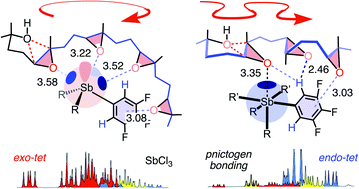
Pnictogen-bond donors are attractive for use in catalysis because of deep σ holes, high multivalency, rich hypervalency, and chiral binding pockets. We here report natural product inspired epoxide-opening polyether cyclizations catalyzed by fluoroarylated Sb(V) > Sb(III) > Bi > Sn > Ge. The distinctive characteristic found for pnictogen-bonding catalysis is the breaking of the Baldwin rules, that is selective endo cyclization into the trans-fused ladder oligomers known from the brevetoxins. Moreover, tris(3,4,5-trifluorophenyl)stibines and their hypervalent stiborane catecholates afford different anti-Baldwin stereoselectivity. Lewis (SbCl3), Brønsted (AcOH) and π acids fail to provide similar access to these forbidden rings. Like hydrogen-bonding catalysis differs from Brønsted acid catalysis, pnictogen-bonding catalysis thus emerges as the supramolecular counterpart of covalent Lewis acid catalysis.


 Please wait while we load your content...
Please wait while we load your content...