Dialumenes – aryl vs. silyl stabilisation for small molecule activation and catalysis†
Abstract
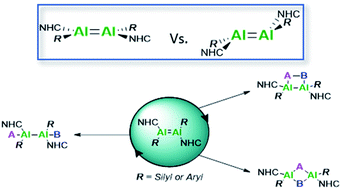
Main group multiple bonds have proven their ability to act as transition metal mimics in the last few decades. However, catalytic application of these species is still in its infancy. Herein we report the second neutral NHC-stabilised dialumene species by use of a supporting aryl ligand (3). Different to the trans-planar silyl-substituted dialumene (3Si), compound 3 features a trans-bent and twisted geometry. The differences between the two dialumenes are explored computationally (using B3LYP-D3/6-311G(d)) as well as experimentally. A high influence of the ligand's steric demand on the structural motif is revealed, giving rise to enhanced reactivity of 3 enabled by a higher flexibility in addition to different polarisation of the aluminium centres. As such, facile activation of dihydrogen is now achievable. The influence of ligand choice is further implicated in two different catalytic reactions; not only is the aryl-stabilised dialumene more catalytically active but the resulting product distributions also differ, thus indicating the likelihood of alternate mechanisms simply through a change of supporting ligand.


 Please wait while we load your content...
Please wait while we load your content...