Transforming colloidal Cs4PbBr6 nanocrystals with poly(maleic anhydride-alt-1-octadecene) into stable CsPbBr3 perovskite emitters through intermediate heterostructures†
Abstract
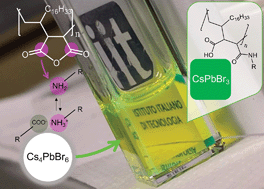
The preparation of strongly emissive CsPbBr3 perovskite nanocrystals with robust surface passivation is a challenge in the field of lead halide perovskite nanomaterials. We report an approach to prepare polymer-capped CsPbBr3 perovskite nanocrystals by reacting oleylammonium/oleate-capped Cs4PbBr6 nanocrystals with poly(maleic anhydride-alt-1-octadecene) (PMAO). PMAO contains succinic anhydride units that are reactive towards the oleylamine species present on the Cs4PbBr6 nanocrystals' surface and produces polysuccinamic acid, which, in turn, triggers the Cs4PbBr6 to CsPbBr3 conversion. The transformation occurs through the formation of Cs4PbBr6–CsPbBr3 heterostructures as intermediates, which are captured because of the mild reactivity of PMAO and are investigated by high-resolution electron microscopy. The Cs4PbBr6–CsPbBr3 heterostructures demonstrate a dual emission at cryogenic temperature with an indication of the energy transfer from Cs4PbBr6 to CsPbBr3. The fully-transformed CsPbBr3 NCs have high photoluminescence quantum yield and enhanced colloidal stability, which we attribute to the adhesion of polysuccinamic acid to the NC surface through its multiple functional groups in place of oleate and alkylammonium ligands. The PMAO-induced transformation of Cs4PbBr6 NCs opens up a strategy for the chemical modification of metal halide NCs initially passivated with nucleophilic amines.
- This article is part of the themed collection: Celebrating five years of ChemRxiv


 Please wait while we load your content...
Please wait while we load your content...