In situ monitoring of mechanochemical synthesis of calcium urea phosphate fertilizer cocrystal reveals highly effective water-based autocatalysis†
Abstract
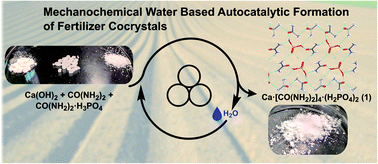
Using the mechanosynthesis of the calcium urea phosphate fertilizer cocrystal as a model, we provide a quantitative investigation of chemical autocatalysis in a mechanochemical reaction. The application of in situ Raman spectroscopy and synchrotron X-ray powder diffraction to monitor the reaction of urea phosphate and either calcium hydroxide or carbonate enabled the first quantitative and in situ study of a mechanochemical system in which one of the products of a chemical reaction (water) mediates the rate of transformation and underpins positive feedback kinetics. The herein observed autocatalysis by water generated in the reaction enables reaction acceleration at amounts that are up to 3 orders of magnitude smaller than in a typical liquid-assisted mechanochemical reaction.
- This article is part of the themed collection: Most popular 2019-2020 catalysis articles


 Please wait while we load your content...
Please wait while we load your content...