The antioxidant activity of natural diterpenes: theoretical insights†
Abstract
Diterpenes that were isolated from Crossopetalum gaumeri (Loes.) Lundell (Celastraceae) plants are reported to exhibit a range of biological activities, in particular as radical scavengers. Thus further insight into the antioxidant activity of diterpenes in physiological environments is much needed but not studied yet. In this study, the antioxidant activity of nine natural diterpenes was evaluated using kinetic and thermodynamic calculations. It was found that the sequential proton loss electron transfer (SPLET) mechanism is favored in polar environments, whereas formal hydrogen transfer (FHT) is the main pathway for the radical scavenging of these diterpenes in the gas phase as well as in lipid media. The rate constants for the HOO˙ radical scavenging of these compounds in the gas phase, polar and nonpolar solvents are in the range of 2.29 × 10−2 to 4.58 × 107, 9.74 × 10−3 to 1.67 × 108 and 3.54 × 10−5 to 1.31 × 105 M−1 s−1, respectively. 7-Deoxynimbidiol (6), exhibits the highest HOO˙ radical scavenging with koverall = 1.69 × 108 M−1 s−1 and 9.10 × 104 M−1 s−1 in water and pentyl ethanoate solvents, respectively, that is about 1300 times higher than that of Trolox in polar environments. It is thus a promising natural antioxidant in physiological environments.
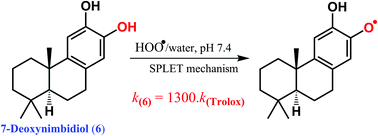


 Please wait while we load your content...
Please wait while we load your content...