In situ Raman and FTIR spectroscopic study on the formation of the isomers MIL-68(Al) and MIL-53(Al)†
Abstract
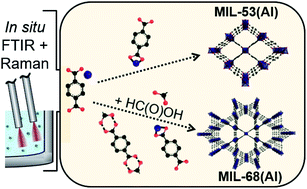
The topological metal–organic framework isomers MIL-53 and MIL-68 form from similar educts but differ in their pore geometries. They have been known for several years, but their synthesis is always reported separately. In consequence, the underlying mechanism and decisive synthesis parameters leading to the formation of either MIL-53 or MIL-68 are not understood. The present study shows how to induce the formation of MIL-68(Al) rather than MIL-53(Al) at low synthesis temperatures in N,N-dimethylformamide (DMF) using a modulated synthesis approach. MIL-68(Al) is identified as the intermediate product of formic acid modulated synthesis, which converts into the thermodynamically stable MIL-53(Al) product at longer synthesis times. The interactions of formic acid with the synthesis precursors responsible for inducing MIL-68(Al) formation are investigated with in situ Raman and FTIR spectroscopy. In contrast to the commonly assumed modulation mechanism of competitive coordination of linker and modulator with the metal node, formic acid is shown to form hydrogen bonds via the carboxylic group of the terephthalic acid (H2BDC) linker, slowing prenucleation building unit and subsequent crystal growth. MIL-68(Al) formation is favored by the combination of a deficiency of terephthalic acid in solution and a slow MOF growth rate. Dissolved H2BDC in solution is proposed to hinder MIL-68(Al) formation by serving as a molecular template for the rhombic MIL-53(Al) pore channels.


 Please wait while we load your content...
Please wait while we load your content...