Synthesis, characterization and antifungal activities of eco-friendly palladium nanoparticles†
Abstract
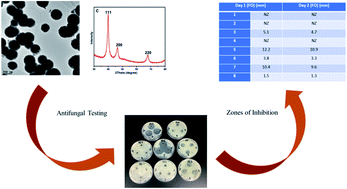
Palladium is a versatile catalyst, but the synthesis of palladium nanoparticles (PdNPs) is usually attained at a high temperature in the range of 160 °C to 200 °C using toxic reducing agents such as sodium borohydride. We report the synthesis of PdNPs using a low-cost and environmentally-friendly route at ambient temperatures. Quercetin diphosphate (QDP), a naturally-derived flavonoid, was employed as a reducing, capping, and stabilizing agent. The effect of temperature was optimized to produce perfectly spherical PdNP nanoparticles with sizes ranging from 0.1 to 0.3 microns in diameter. At relatively higher concentration of QDP, significantly smaller particles were produced with a size distribution of 1–7 nm. Perfectly spherical PdNP nanoparticles are a rare occurrence, especially under ambient room temperature conditions with fast reaction time. The formation of the nanoparticles was confirmed using UV-vis, TEM, EDS, and XRD. HRTEM demonstrated the lattice structure of the PdNPs. The synthesized PdNPs were also tested for their antifungal properties against Colletotrichum gloeosporioides and Fusarium oxysporum. Results showed that the size of the PdNPs played a critical role in their antifungal activity. However, for F. oxysporum, other factors beyond size could affect the antifungal activity including fine-scale, nutrient composition, and target organisms.


 Please wait while we load your content...
Please wait while we load your content...