On the molecular architectures of siloxane coordination compounds: (re-)investigating the coordination of the cyclodimethylsiloxanes Dn (n = 5–8) towards alkali metal ions†
Abstract
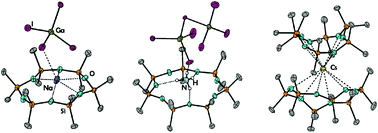
In this study we present a (re-)investigation into cyclodimethylsiloxanes in relation to silyl-ether bonding towards alkali metal ions. We demonstrate that all (non-radioactive) alkali metal ions can be incorporated into Dn cyclosiloxane frameworks (D = (SiMe2O), n = 5–8), employing appropriate cation–anion combinations. Starting with the Li+ cation we were able to observe the coordination of D5 with Li+ based on a suitable X-ray structure after reacting D5 with LiI and GaI3. Due to template effects, the dinuclear coordination compound [Li2(D5)(D6)(GaI4)2] (1) was obtained. The direct reaction of D6 with LiI and GaI3 yields [Li(D6)GaI4] (2) in quantitative yield. Na+ ions could be trapped in D6 and D7 ligand moieties after the conversion of NaI, GaI3, and the respective siloxane. The molecular structure of [Na(D6)GaI4] (3) reveals a six-fold coordinated Na+ ion, which is located on top of the siloxane D6. In the case of [Na(D7)(DCM)GaI4] (4), the larger ligand D7 provides 15-crown-5-like geometry in which the sodium ion is coordinated by the ligand in a coplanar fashion and further saturated by the solvent DCM (DCM = dichloromethane). The K+ ion was bound within the D7 ligand in a similar manner and [K(D7)(DCM)GaI4] (5) could be characterized. Due to the resemblance of NH4+ to K+, this cation was also employed for complex formation. Counterintuitively, we were able to synthesize and characterize the first ever non-metal-cyclosiloxane coordination compound. After the conversion of D6 with NH4I and GaI3, the compound [NH4(D7)][Ga2I7] (6) was obtained. The ammonium cation favors D7 coordination over D6, and the willing formation of hydrogen bonding in such a siloxane moiety was realized. As these compounds could be obtained, we also tested the limits of silyl-ether bonding. Therefore, we reacted D8 with in situ generated Rb[GaI4] and Cs[GaI4]. In the case of Rb+, we could cumbersomely characterize [Rb(D8)(DCM)GaI4] (7) via an X-ray structure, as well as by means of mass spectrometry, but the compound starts decomposing readily in solution. The reaction with the Cs+ salt failed. To obtain meaningful spectroscopic data from a Rb+ compound and to somehow obtain a Cs+ complex, we employed the weakly coordinating anion [AlF]− (AlF− = [Al{OC(CF3)3}4]−). The conversion of M[AlF] then yielded 1∞[M(D8)ALF] (M = Rb: 8; M = Cs+: 9) in the solid state. Both compounds 8 and 9 were fully characterized. Finally, we aimed at synthesizing 2 : 1 complexes of such siloxanes. The reactions of excess D5 with K[AlF] and D6 with Cs[AlF] turned out to be expedient and, in the forms of [K(D5)2][AlF] (10) and [Cs(D6)2][AlF] (12), the first ever sandwich-type complexes observed bearing a cyclosiloxane ligand were characterized.


 Please wait while we load your content...
Please wait while we load your content...