Cellular localisation of structurally diverse diphenylacetylene fluorophores†
Abstract
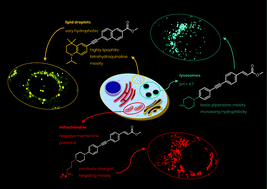
Fluorescent probes are increasingly used as reporter molecules in a wide variety of biophysical experiments, but when designing new compounds it can often be difficult to anticipate the effect that changing chemical structure can have on cellular localisation and fluorescence behaviour. To provide further chemical rationale for probe design, a series of donor–acceptor diphenylacetylene fluorophores with varying lipophilicities and structures were synthesised and analysed in human epidermal cells using a range of cellular imaging techniques. These experiments showed that, within this family, the greatest determinants of cellular localisation were overall lipophilicity and the presence of ionisable groups. Indeed, compounds with high log D values (>5) were found to localise in lipid droplets, but conversion of their ester acceptor groups to the corresponding carboxylic acids caused a pronounced shift to localisation in the endoplasmic reticulum. Mildly lipophilic compounds (log D = 2–3) with strongly basic amine groups were shown to be confined to lysosomes i.e. an acidic cellular compartment, but sequestering this positively charged motif as an amide resulted in a significant change to cytoplasmic and membrane localisation. Finally, specific organelles including the mitochondria could be targeted by incorporating groups such as a triphenylphosphonium moiety. Taken together, this account illustrates a range of guiding principles that can inform the design of other fluorescent molecules but, moreover, has demonstrated that many of these diphenylacetylenes have significant utility as probes in a range of cellular imaging studies.
- This article is part of the themed collection: Chemical Biology in OBC


 Please wait while we load your content...
Please wait while we load your content...