Solvent induced mononuclear and dinuclear mixed ligand Cu(ii) complex: structural diversity, supramolecular packing polymorphism and molecular docking studies†
Abstract
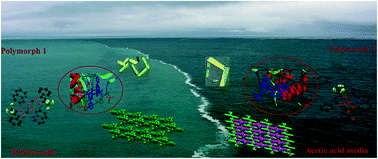
A novel mixed ligand Cu(II) complex of 4,4,4-trifluoro-1-phenyl-1,3-butanedione and 8-hydroxyquinoline was synthesized and spectroscopically characterized. The phenolate oxygen bridged dinuclear (polymorph 1) and solvent-induced mononuclear (polymorph 2) forms were obtained using DMSO and acetic acid solvents. The solvato supramolecular isomeric forms (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) and P21/a) of the complex are confirmed by the single crystal X-ray diffraction method. In polymorph 1, a hydrogen-bonded one-dimensional network is constructed by C–H⋯O contacts, whereas, in polymorph 2, a two-dimensional ladder-like architecture is formed by the interplay of C–H⋯O, C–H⋯F and F⋯F contacts. The vital role of distinct non-covalent interactions in the establishment of a unique structural topology is analysed by Hirshfeld surfaces, 2D fingerprint plots, QTAIM and the NCI index model. The anisotropies in the three-dimensional structural topology are visualized through their energy frameworks and evidenced by their interaction energies. The electronic structures of the polymorphs were optimized by density functional theory calculations to explore their coordination stability and chemically reactive parameters. Polymorph 1 exhibits potential antibacterial activity against methicillin-resistant Staphylococcus aureus with a minimum inhibitory concentration of 8 μg mL−1. Further, polymorphs were docked to penicillin-binding protein 2, where polymorph 1 had a binding score of −10.0 kcal mol−1, greatly supporting the in vitro results.
and P21/a) of the complex are confirmed by the single crystal X-ray diffraction method. In polymorph 1, a hydrogen-bonded one-dimensional network is constructed by C–H⋯O contacts, whereas, in polymorph 2, a two-dimensional ladder-like architecture is formed by the interplay of C–H⋯O, C–H⋯F and F⋯F contacts. The vital role of distinct non-covalent interactions in the establishment of a unique structural topology is analysed by Hirshfeld surfaces, 2D fingerprint plots, QTAIM and the NCI index model. The anisotropies in the three-dimensional structural topology are visualized through their energy frameworks and evidenced by their interaction energies. The electronic structures of the polymorphs were optimized by density functional theory calculations to explore their coordination stability and chemically reactive parameters. Polymorph 1 exhibits potential antibacterial activity against methicillin-resistant Staphylococcus aureus with a minimum inhibitory concentration of 8 μg mL−1. Further, polymorphs were docked to penicillin-binding protein 2, where polymorph 1 had a binding score of −10.0 kcal mol−1, greatly supporting the in vitro results.


 Please wait while we load your content...
Please wait while we load your content...