Galvanic replacement of liquid metal Galinstan with copper for the formation of photocatalytically active nanomaterials†
Abstract
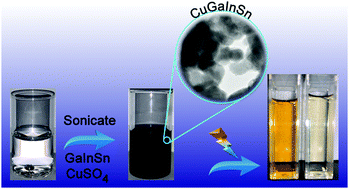
The fabrication of low cost and highly active photocatalysts for the degradation of synthetic dyes that are effective under visible light is an ongoing challenge. Here, the synthesis of a multi-elemental CuGaInSn nanocomposite by sonication assisted galvanic replacement of liquid metal Galinstan (GaInSn) in copper sulphate solution is demonstrated as a method to produce a photocatalyst for dye degradation. It was found that the copper salt concentration and synthesis solution pH determined the composition of the final nanomaterial by minimizing surface oxidation of the catalyst and promoting the formation of a metallic nanocomposite rather than a metal oxide material. The materials were characterized with X-ray diffraction, X-ray photoelectron spectroscopy, field emission scanning electron microscopy, transmission electron microscopy and electrochemical techniques. This analysis confirmed the presence of a solid mixed phase CuGaInSn nanocomposite which is a potential candidate for environmental remediation applications where the photocatalytic degradation of methyl orange dye was used as a model system. A degradation efficiency of 84% under low power illumination was achieved with a high degree of reusability via immobilization of the catalyst on a solid support, while a degradation efficiency of 92% was achieved under solar simulated conditions.


 Please wait while we load your content...
Please wait while we load your content...