Perturbing the AIEE activity of pyridine functionalized α-cyanostilbenes with donor substitutions: an experimental and DFT study†
Abstract
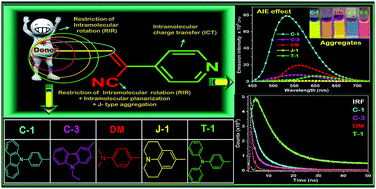
A series of pyridine functionalized α-cyanostilbenes bearing different amino group donors, carbazole–phenyl (C-1), N-ethyl carbazole (C-3), dimethylamino (DM), julolidine (J-1) and triphenylamine (T-1), were synthesized, and their impact on the aggregation propensity of the α-cyanostilbenes was investigated. Distinct aggregation-induced emission (AIE) features have been obtained in water, where the emission intensity varied in the following order: C-1 > DM > C-3 > T-1 > J-1. The formation of aggregates was substantiated by fluorescence lifetime decay profiles and their average lifetime correlated with the rigidity of the molecular framework. The contributing factors behind the AIE phenomena are the bulkiness of the donor segments, their donating strength, planarity of the molecular framework, the distance between the α-cyano unit and the donors and its interaction with neighboring units. Based on the experimental observations from this study and previous research reports, we have used DFT and TDDFT methods to evaluate the donor–AIEE property relationship. Cyclic voltammetry techniques were used to examine the redox properties of the compounds. SEM and DLS studies provided us with a detailed picture of the morphology and hydrodynamic radii of the aggregates, respectively. The results obtained from this study would be helpful to understand the substituent dependent aggregation propensity of α-cyanostilbenes and provide design guidelines toward the development of AIE active materials for various optoelectronic and bioimaging applications.


 Please wait while we load your content...
Please wait while we load your content...