Insights into the mechanism of the formation of noble metal nanoparticles by in situ NMR spectroscopy†
Abstract
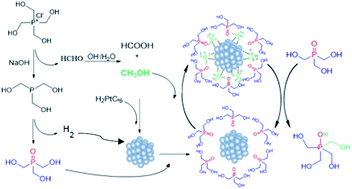
High-resolution solution Nuclear Magnetic Resonance (NMR) spectroscopy has been used to gain insights into the mechanism of the formation of gold, platinum and gold–platinum alloyed nanoparticles using metal precursors and tetrakis(hydroxymethyl)phosphonium chloride (THPC) as starting materials. THPC is widely used in nanochemistry as a reductant and stabilizer of nanoparticles, however the identity of the species responsible for each role is unknown. The multinuclear study of the reaction media by NMR spectroscopy allowed us to elucidate the structure of all the compounds that participate in the transformation from the metal salt precursor to the reduced metal that forms the nanoparticle, thus clarifying the controversy found in the literature regarding the formation of THPC-based compounds. The progress of the reaction was monitored from the initial moments of the synthesis to the end of the reaction and after long periods of time. Insights into the dual role of THPC were gained, identifying methanol and hydrogen as the actual reducing agents, and tris(hydroxymethyl)phosphine oxide (THPO) as the real stabilizing agent. Finally, the different stabilities of gold and platinum nanoparticles can be attributed to the different catalytic activities of the metals.


 Please wait while we load your content...
Please wait while we load your content...