Selective metathesis synthesis of MgCr2S4 by control of thermodynamic driving forces†
Abstract
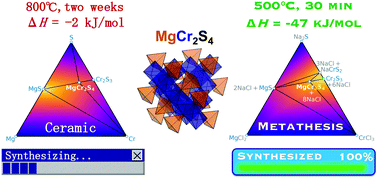
MgCr2S4 thiospinel is predicted to be a compelling Mg-cathode material, but its preparation via traditional solid-state synthesis methods has proven challenging. Wustrow et al. [Inorg. Chem., 2018, 57, 14] found that the formation of MgCr2S4 from MgS + Cr2S3 binaries requires weeks of annealing at 800 °C with numerous intermediate regrinds. The slow reaction kinetics of MgS + Cr2S3 → MgCr2S4 can be attributed to a miniscule thermodynamic driving force of ΔH = −2 kJ mol−1. Here, we demonstrate that the double ion-exchange metathesis reaction, MgCl2 + 2NaCrS2 → MgCr2S4 + 2NaCl, has a reaction enthalpy of ΔH = −47 kJ mol−1, which is thermodynamically driven by the large exothermicity of NaCl formation. Using this metathesis reaction, we successfully synthesized MgCr2S4 nanoparticles (<200 nm) from MgCl2 and NaCrS2 precursors in a KCl flux at 500 °C in only 30 minutes. NaCl and other metathesis byproducts are then easily washed away by water. We rationalize the selectivity of MgCr2S4 in the metathesis reaction from the topology of the DFT-calculated pseudo-ternary MgCl2–CrCl3–Na2S phase diagram. Our work helps to establish metathesis reactions as a powerful alternative synthesis route to inorganic materials that have otherwise small reaction energies from conventional precursors.
- This article is part of the themed collection: Materials Horizons Emerging Investigators Series 2020/2021


 Please wait while we load your content...
Please wait while we load your content...