Diketopyrrolopyrrole linked porphyrin dimers for visible-near-infrared photoresponsive nonfullerene organic solar cells†
Abstract
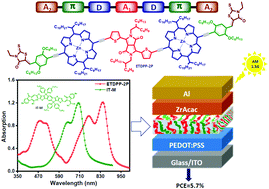
Two new A2–π–D–A1–D–π–A2 structural porphyrin small molecules denoted as TDPP-2P and ETDPP-2P are constructed from dimeric porphyrin centers, in which the two porphyrin moieties are linked by either the electron deficient 3,6-di(thiophen-2-yl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione (TDPP) for the former, or diethynyl substituted TDPP for the latter, further symmetrically π-extended with electron withdrawing rhodanine units through phenylenethynylene π-bridges. For both, 2-butyloctyl and 1-octylundecyl branched alkyl chains are attached to TDPP and porphyrin moieties, respectively, to improve their solubility and self-assembly properties. Compared to TDPP-2P, ETDPP-2P shows a broader absorption in the near-infrared (NIR) range over 1000 nm due to the enhanced coplanarity of the dimeric porphyrin center. Using IT-M as the acceptor, the blend film ETDPP-2P:IT-M accomplishes a panchromatic photoresponse from 300–900 nm, and as a result, the device exhibits a prominent PCE of 5.69%, whereas, the film TDPP-2P:IT-M shows a lower PCE of 4.12% due to the lack of NIR photoresponses. These primary results open up a promising avenue for constructing porphyrin-based all small molecule organic solar cells with a panchromatic photoresponse.
- This article is part of the themed collection: Editor’s Choice: Organic Photovoltaics


 Please wait while we load your content...
Please wait while we load your content...