Ferroelectric surface photovoltage enhancement in chromium-doped SrTiO3 nanocrystal photocatalysts for hydrogen evolution†
Abstract
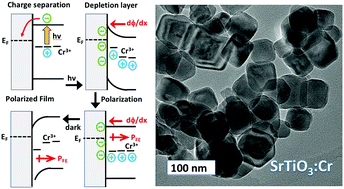
Chromium-doped SrTiO3 nanocrystals of perovskite structure type and 45 nm (±15 nm) edge lengths were obtained by hydrothermal synthesis in water from titanium oxide, strontium hydroxide, and chromium(III) nitrate. According to XPS, the majority of the surface chromium (68.3%) is present in the 3+ state and the remainder (32.2%) in the 6+ state. Optical spectroscopy confirms a broad absorption at 2.3–2.9 eV from Cr(3+) dopant states, in addition to the 3.2 eV band edge of the SrTiO3 host. After modification with Pt nanoparticles, Cr-doped SrTiO3 nanocrystals catalyze photochemical H2 evolution from aqueous methanol under visible light illumination (>400 nm) and with an apparent quantum yield of 0.66% at 435 nm. According to surface photovoltage spectroscopy (SPS), Cr-doped SrTiO3 nanocrystals deposited onto gold substrates are n-type and have an effective band gap of 1.75 eV. SPS and transient illumination experiments at 2.50 eV reveal an anomalous surface photovoltage that increases with prior light exposure to values of up to −6.3 V. This photovoltage is assigned to ferroelectric polarization of the material in the space charge layer at the Au/SrTiO3:Cr interface. The polarization is stable for 24 h in vacuum but disappears after 12 h when samples are stored in air. The electric polarizability of SrTiO3:Cr is confirmed when films are exposed to static electric fields (1.20 MV m−1) in a fixed capacitor configuration. The discovery of a ferroelectric effect in Cr-doped SrTiO3 could be significant for the development of improved photocatalysts for the conversion of solar energy into fuel.


 Please wait while we load your content...
Please wait while we load your content...