Degradation of a chloroacetanilide herbicide in natural waters using UV activated hydrogen peroxide, persulfate and peroxymonosulfate processes†
Abstract
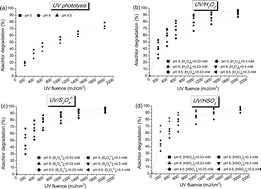
In water treatment, the application of advanced oxidation processes (AOPs) which involve the generation of not only hydroxyl but also sulfate radicals has recently attracted increasing attention worldwide. To the best of our knowledge, to date, the application of ultraviolet (UV) activated persulfate and peroxymonosulfate (UV/S2O82− and UV/HSO5−) has not been comprehensively studied for chloroacetanilide herbicide degradation. In our study, this process was investigated under different conditions and compared to the more conventional UV/H2O2 process. Alachlor was investigated due to its toxic properties, and the threat it poses to aquatic environments and human health, which led to its classification as a priority pollutant by the European Water Framework Directive. The alachlor degradation kinetics of different UV-based AOPs were highly dependent on the pH and the water matrix characteristics. The UV/S2O82− and UV/H2O2 processes proved to be most effective under acidic conditions (pH 5), while the UV/HSO5− process showed the highest alachlor degradation efficacy under basic conditions (pH > 8). The groundwater matrix had a greater impact on the AOPs than the surface water. PCA analysis was applied and a predictive model of alachlor degradation by UV-based AOPs was proposed. Alachlor degradation pathways were also evaluated together with NOM transformation during the applied treatments. The obtained results indicate that the more novel UV/S2O82− and UV/HSO5− processes show significant potential for herbicide degradation during water treatment, especially when the raw water is highly loaded with hydrophobic NOM.


 Please wait while we load your content...
Please wait while we load your content...