Isotope fractionation (2H/1H, 13C/12C, 37Cl/35Cl) in trichloromethane and trichloroethene caused by partitioning between gas phase and water†
Abstract
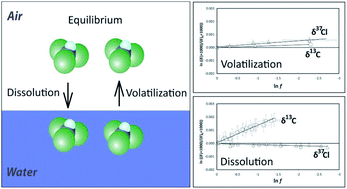
Transfer of organic compounds between aqueous and gaseous phases may change the isotopic composition which complicates the isotopic characterization of sources and transformation mechanisms in environmental samples. Studies investigating kinetic phase transfer of compounds dissolved in water (volatilization) are scarce, even though it presents an environmentally very relevant phase transfer scenario. In the current study, the occurrence of kinetic isotope fractionation (2H/1H, 13C/12C, 37Cl/35Cl) was investigated for two volatile organic compounds (trichloroethene, TCE and trichloromethane, TCM) during volatilization from water and gas-phase dissolution in water. In addition, experiments were also carried out at equilibrium conditions. The results indicated that volatilization of trichloromethane and trichloroethene from water, in contrast to pure phase evaporation, only caused small (chlorine) or negligible (hydrogen, carbon) isotope fractionation whereas for dissolution in water significant carbon isotope effects were found. At equilibrium conditions, hydrogen and carbon isotopes showed significant differences between dissolved and gaseous phase whereas small to insignificant differences were measured for chlorine isotopes. The results confirm the hypothesis that isotope effects during volatilization of organics from water are caused by transport inhibition in the aqueous phase. The consideration of gas-phase diffusion and vapor pressure isotope effects (Craig–Gordon model) could not reproduce the measured isotopic data. Overall, this study provides an overview of the most common kinetic and equilibrium partitioning scenarios and reports associated isotope effects. As such it illustrates under which environmental conditions isotopic signatures of chlorinated volatile organics may change, or remain constant, during transfer between surface waters and air.
- This article is part of the themed collection: Halogenated (semi)volatile organic compounds (“X(S)VOCs”)


 Please wait while we load your content...
Please wait while we load your content...