Abstract
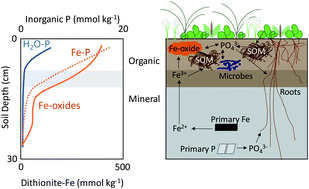
Phosphorus (P) is a limiting or co-limiting nutrient to plants and microorganisms in diverse ecosystems that include the arctic tundra. Certain soil minerals can adsorb or co-precipitate with phosphate, and this mineral-bound P provides a potentially large P reservoir in soils. Iron (Fe) oxyhydroxides have a high capacity to adsorb phosphate; however, the ability of Fe oxyhydroxides to adsorb phosphate and limit P bioavailability in organic tundra soils is not known. Here, we examined the depth distribution of soil Fe and P species in the active layer (<30 cm) of low-centered and high-centered ice-wedge polygons at the Barrow Environmental Observatory on the Alaska North Slope. Soil reservoirs of Fe and P in bulk horizons and in narrower depth increments were characterized using sequential chemical extractions and synchrotron-based X-ray absorption spectroscopy (XAS). Organic horizons across all polygon features (e.g., trough, ridge, and center) were enriched in extractable Fe and P relative to mineral horizons. Soil Fe was dominated by organic-bound Fe and short-range ordered Fe oxyhydroxides, while soil P was primarily associated with oxides and organic matter in organic horizons but apatite and/or calcareous minerals in mineral horizons. Iron oxyhydroxides and Fe-bound inorganic P (Pi) were most enriched at the soil surface and decreased gradually with depth, and Fe-bound Pi was >4× greater than water-soluble Pi. These results demonstrate that Fe-bound Pi is a large and ecologically important reservoir of phosphate. We contend that Fe oxyhydroxides and other minerals may regulate Pi solubility under fluctuating redox conditions in organic surface soils on the arctic tundra.
- This article is part of the themed collection: Cryosphere Chemistry


 Please wait while we load your content...
Please wait while we load your content...
