Reaction of GaIIIClPc, SnIVCl2TPP and BIIIClSubPc with cyanide anions: reduction of macrocycles vs. formation of cyano-containing macrocyclic anions†
Abstract
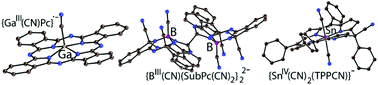
The reaction of GaIIIClPc, SnIVCl2TPP and BIIIClSubPc containing phthalocyanine (Pc), tetraphenylporphyrin (TPP) and subphthalocyanine (SubPc) macrocycles with cyanide in the presence of cryptand[2.2.2] under anaerobic conditions yields crystalline salts in which cyano anions substitute chloride anions at GaIII, SnIV or BIII, as well as reducing the macrocycles or adding one or two CN− to them. The reaction of GaIIICl(Pc2−) with CN− yields {crypt(K+)}{GaIIICN(Pc˙3−)}˙−·0.5C6H4Cl2 (1) in which the Pc2− macrocycle is reduced to Pc˙3−. Such reduction could probably occur through the addition of CN− to Pc2− forming {GaIII(CN)[Pc(CN)]3−}− which can decompose further interacting with an excess of CN−. As a result, Pc˙3− and cyanogene anions are formed. The interaction of SnIVCl2(TPP2−) with CN− is accompanied by the addition of CN− to the meso-carbon atom of porphyrin forming diamagnetic TPP(CN)3− macrocycles in {crypt(K+)}{SnIV(CN)2[TPP(CN)]3−}− (2). Salt 2 shows a strong NIR absorption band with the maximum at 854 nm whose intensity is comparable with that of the Soret band. The interaction of BIIICl(SubPc2−) with three equivalents of CN− is accompanied by the addition of two CN− to carbon atoms of SubPc2− closest to meso-nitrogen atoms forming {BIII(CN)[SubPc(CN)2]4−}2−. Most probably these dianions transfer electrons to C6H4Cl2 producing the {BIII(CN)[SubPc(CN)2]˙3−}˙− radical anions which form σ-bonded diamagnetic dianions in {crypt(K+)}2{BIII(CN)[SubPc(CN)2]}22−·3C6H4Cl2 (3). The remaining carbon atom closest to the meso-nitrogen atom is involved in this dimerization. According to the calculations, the energy of the C–CN bond is minimal for {GaIII(CN)[Pc(CN)]3−}− enabling further transformation of these anions to {GaIIICN(Pc˙3−)}˙− in 1, whereas cyano-containing anions in 2 and 3 with higher energy of this bond are stable towards the elimination of CN. Optical and magnetic properties of 1–3 together with their crystal and molecular structures are presented. The possible ways of the formation of 1–3 are discussed based on DFT calculations.


 Please wait while we load your content...
Please wait while we load your content...