Recent developments in the control of selectivity in hydrogenation reactions by confined metal functionalities†
Abstract
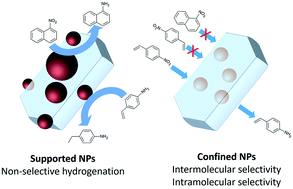
Confining metal active species in the voids of porous solid matrices such as zeolites, metal–organic frameworks (MOFs), and carbon nanotubes (CNTs) can bring fascinating key advantages in the field of selective hydrogenation reactions. Confined metal species act as intermolecular selective catalysts capable of discriminating reagents based on their molecular size and shape. They also exhibit intramolecular selectivity by converting one or more functional groups selectively in the presence of others. In this review, we present a comprehensive overview of the different synthetic methods for confining active metal species in the voids of zeolites, MOFs, CNTs, and other porous structures. We then emphasize the strong influence of metal confinement on steering catalytic selectivity in a wide range of selective hydrogenation reactions. Finally, we share our opinion on the different synthesis methods for potential practical applications and on future research directions.


 Please wait while we load your content...
Please wait while we load your content...