Structural dynamics in Ni–Fe catalysts during CO2 methanation – role of iron oxide clusters†
Abstract
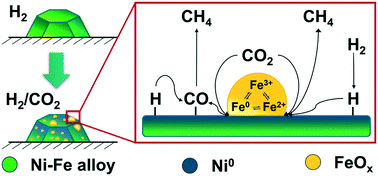
Bimetallic Ni–Fe catalysts show great potential for CO2 methanation concerning activity, selectivity and long-term stability even under transient reaction conditions as required for Power-to-X applications. Various contrary suggestions on the role of iron in this system on CO2 activation have been proposed, hence, its actual task remained still unclear. In this study, we used X-ray absorption spectroscopy (XAS) combined with X-ray diffraction (XRD), XAS in combination with modulation excitation spectroscopy (MES) and density functional theory (DFT) to shed detailed light on the role of iron in a bimetallic Ni–Fe based CO2 methanation catalyst. During catalyst activation we observed a synergistic effect between nickel and iron that led to higher fractions of reduced nickel compared to a monometallic Ni-based catalyst. By XAS–XRD combined with DFT, we found formation of FeOx clusters on top of the metal particles. Modulation excitation coupled XAS data complemented with DFT calculations provided evidence of a Fe0 ⇌ Fe2+ ⇌ Fe3+ redox mechanism at the interface of these FeOx clusters. This may promote CO2 dissociation. This is the first time that this highly dynamic role of iron has been experimentally confirmed in bimetallic Ni–Fe based catalysts with respect to CO2 activation during the methanation reaction and may also be at the origin of better performance of other CO2-hydrogenation catalysts. The insight into the structural surface changes reported in this study show the dynamics of the Fe–Ni system and allow the development of realistic surface models as basis for CO2 activation and possible intermediates in these bimetallic systems.


 Please wait while we load your content...
Please wait while we load your content...