Investigating methane dry reforming on Ni and B promoted Ni surfaces: DFT assisted microkinetic analysis and addressing the coking problem†
Abstract
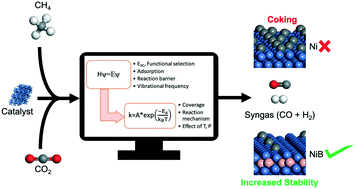
Dry reforming of methane (DRM) has incited significant academic and industrial attention in the past couple of decades. Although Ni-based catalysts have shown good activity for DRM, deactivation due to carbon formation is a serious concern. Several strategies are proposed, including doping by other metals and metalloids, to improve the stability of Ni, and hence we investigated boron-doped Ni (NiB) as a potential catalyst for DRM. In this work, we combined DFT and microkinetic modeling to identify the dominant reaction pathways and kinetically relevant steps of the DRM reaction system on Ni and NiB surfaces. We considered a detailed reaction network involving multiple CO2 and CH4 dissociation routes, side reactions (H2O formation, Boudouard reaction) and desorption of products on both surfaces. Our DFT calculations suggest that both Ni and NiB share similar CO2 (direct dissociation to CO*) and CH4 (sequential dehydrogenation to C*) activation routes. Compared to Ni, the CO2 activation barrier on NiB is higher by 44 kJ mol−1 but the barriers in CH4 activation routes are significantly lower. Subsequently, the DFT-calculated energies were used to derive kinetic rate constants of the elementary reaction steps. These rate constants were employed to develop a microkinetic model of the DRM process over Ni (111) and NiB catalysts. At low CH4 and CO2 partial pressures (<10 kPa) and low total reaction pressure (1 bar) both CH4 dissociative adsorption and CO2 adsorption dominate the overall reaction rate. However, at high total reaction pressure (10 bar) and low reaction temperatures (873 and 973 K) only CH4 dissociation dominates the overall rate. As the temperature is increased (1073 K), it is dominated by the CH* oxidation. It was observed that the dominant pathway on Ni causes carbon formation on the catalyst surface. Whereas the dominant reaction pathway on NiB includes 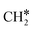 oxidation, that prevents carbon formation, making it a more stable catalyst. Furthermore, the forward rate constant of Boudouard reaction
oxidation, that prevents carbon formation, making it a more stable catalyst. Furthermore, the forward rate constant of Boudouard reaction 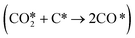 was significantly higher on NiB and hence the carbon formed is consumed at a faster rate. Thus, we believe that NiB is a potential catalyst that can resist deactivation during the DRM process.
was significantly higher on NiB and hence the carbon formed is consumed at a faster rate. Thus, we believe that NiB is a potential catalyst that can resist deactivation during the DRM process.


 Please wait while we load your content...
Please wait while we load your content...