Semiconductor nanocrystals for small molecule activation via artificial photosynthesis
Abstract
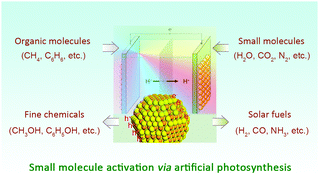
Facile activation and conversion of small molecules (e.g., H2O, CO2, N2, CH4, and C6H6) into solar fuels or value-added chemicals under mild conditions is an attractive pathway in dealing with the worldwide appeal of energy consumption and the growing demand of industrial feedstocks. Compared with conventional thermo- or electro-catalytic approaches, the protocol of photocatalysis shines light on green and low-cost storage of sunlight in chemical bonds. For instance, artificial photosynthesis is an effective way to split H2O into molecular O2 and H2, thereby storing solar energy in the form of hydrogen fuel. Because of rational tunability in band gaps, charge-carrier dynamics, exposed active sites and catalytic redox activities by tailoring size, composition, morphology, surface, and/or interface property, semiconductor nanocrystals (NCs) emerge as very promising candidates for photo-induced small molecule activation, including H2O splitting, CO2 reduction, N2 fixation, CH4 conversion and chemical bond formation (e.g., S–S, C–C, C–N, C–P, C–O). In this review, we summarize the recent advances in small molecule activation via artificial photosynthesis using semiconductor NCs, especially those consisting of II–VI and III–V elements. Moreover, we highlight the intrinsic advantages of semiconductor NCs in this field and look into the fabrication of prototype devices for large-scale and sustainable small molecule activation to store solar energy in chemical bonds.
- This article is part of the themed collection: Small molecule activation, from biocatalysis to artificial catalysis


 Please wait while we load your content...
Please wait while we load your content...