Site-specific dynamic nuclear polarization in a Gd(iii)-labeled protein†
Abstract
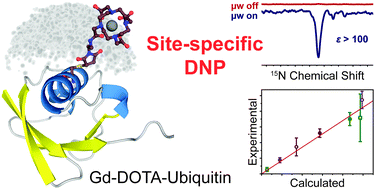
Dynamic nuclear polarization (DNP) of a biomolecule tagged with a polarizing agent has the potential to not only increase NMR sensitivity but also to provide specificity towards the tagging site. Although the general concept has been often discussed, the observation of true site-specific DNP and its dependence on the electron–nuclear distance has been elusive. Here, we demonstrate site-specific DNP in a uniformly isotope-labeled ubiquitin. By recombinant expression of three different ubiquitin point mutants (F4C, A28C, and G75C) post-translationally modified with a Gd3+-chelator tag, localized metal-ion DNP of 13C and 15N is investigated. Effects counteracting the site-specificity of DNP such as nuclear spin–lattice relaxation and proton-driven spin diffusion have been attenuated by perdeuteration of the protein. Particularly for 15N, large DNP enhancement factors on the order of 100 and above as well as localized effects within side-chain resonances differently distributed over the protein are observed. By analyzing the experimental DNP built-up dynamics combined with structural modeling of Gd3+-tags in ubiquitin supported by paramagnetic relaxation enhancement (PRE) in solution, we provide, for the first time, quantitative information on the distance dependence of the initial DNP transfer. We show that the direct 15N DNP transfer rate indeed linearly depends on the square of the hyperfine interaction between the electron and the nucleus following Fermi's golden rule, however, below a certain distance cutoff paramagnetic signal bleaching may dramatically skew the correlation.
- This article is part of the themed collection: 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...