Interpreting time-resolved photoluminescence of perovskite materials†
Abstract
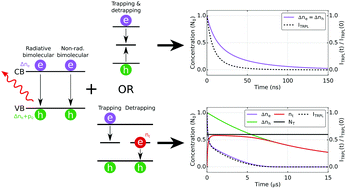
Time-resolved photoluminescence (TRPL) spectroscopy is a powerful technique to investigate excited charge carrier recombinations in semiconductors and molecular systems. The analysis of the TRPL decays of many molecular systems (e.g. molecules and organic materials) is usually fairly straightfoward and can be fitted with an exponential function allowing extraction of the rate constants. Due to the non-excitonic nature of charge carriers in lead halide perovskite materials coupled with the presence of localised trap states in their band-gap, the TRPL of these materials is much more complicated to interpret. Here we discuss two models used in the literature to simulate charge carrier recombinations and TRPL in perovskites. These models consider the bimolecular nature of direct electron–hole recombination but differ in their treatment of trap-mediated recombination with one model describing trapping as a monomolecular process whereas the other as a bimolecular process between free carriers and the available trap states. In comparison, the classical analysis of perovskite TRPL decay curves (using a sum of exponentials) can lead to misinterpretation. Here we offer some recommendations for meaningful measurements of lead halide perovskite thin-films. The fluence dependence as well as charge carrier accumulation due to incomplete depopulation of all photoexcited carriers between consecutive excitation pulses are discussed for both models.


 Please wait while we load your content...
Please wait while we load your content...