The charge reduction rate for multiply charged polymer ions via ion–ion recombination at atmospheric pressure†
Abstract
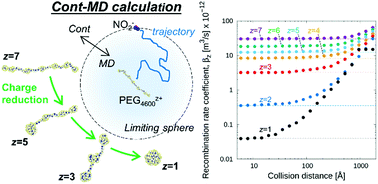
The charge reduction of multiply charged macromolecular ions via recombination with small ions in the gas phase is commonly employed to modulate the charge on macromolecules prior to mass spectrometric and mobility analyses. We employ a recently developed continuum-Molecular Dynamics (MD) calculation approach to determine the recombination rate coefficient of multiply charged (1 to 7 excess positive charged) polyethylene glycol ions (mass of 4600 Da) with smaller singly charged anions, modeled as NO2− ions. The continuum-MD approach accounts explicitly for the influence of the background gas on the recombination process, accounts explicitly for ion translational, vibrational, and rotational motion, and enables recombination rate coefficient calculation in nitrogen near atmospheric pressure, wherein neither low pressure nor high pressure recombination theories are strictly applicable. Continuum-MD simulations yield recombination rate coefficients near 3.9 × 10−14 m3 s−1 for singly charged ions, increasing to 3.0 × 10−11 m3 s−1 for the +7 ion. Pre-existing collision rate coefficient expressions for rigid ions are found to be within a factor of 2–5 of calculations for all charge states, but their use requires knowledge of an appropriate collision distance, which is not well-defined for flexible polymer ions. Continuum-MD-inferred rate coefficients are incorporated into a model of charge reduction, and the charge state distribution versus anion concentration determined with it is compared to charge reduction measurements made via atmospheric pressure differential mobility analysis. Good agreement is observed between simulations and experiments; although results are extremely sensitive to the recombination rate coefficients, experimental results are bound by models utilizing rates within a factor of 2 (0.5–2.0×) of the continuum-MD rates.


 Please wait while we load your content...
Please wait while we load your content...