Surface-orientation- and ligand-dependent quenching of the spin magnetic moment of Co porphyrins adsorbed on Cu substrates†
Abstract
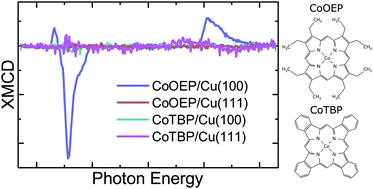
Porphyrin molecules are particularly interesting candidates for spintronic applications due to their bonding flexibility, which allows to modify their properties substantially by the addition or transformation of ligands. Here, we investigate the electronic and magnetic properties of cobalt octaethylporphyrin (CoOEP), deposited on copper substrates with two distinct crystallographic surface orientations, Cu(100) and Cu(111), with X-ray absorption spectroscopy (XAS) and X-ray magnetic circular dichroism (XMCD). A significant magnetic moment is present in the Co ions of the molecules deposited on Cu(100), but it is completely quenched on Cu(111). Heating the molecules on both substrates to 500 K induces a ring-closure reaction with cobalt tetrabenzoporphyrin (CoTBP) as reaction product. In these molecules, the magnetic moment is quenched on both surfaces. Our XMCD and XAS measurements suggest that the filling of the dz2 orbital leads to a non-integer valence state and causes the quench of the spin moments on all samples except CoOEP/Cu(100), where the molecular conformation induces variations to the ligand field that lift the quench. We further employ density functional theory calculations, supplemented with on-site Coulomb correlations (DFT+U), to study the adsorption of these spin-bearing molecules on the Cu substrates. Our calculations show that charge transfer from the Cu substrates as well as charge redistribution within the Co 3d orbitals lead to the filling of the Co minority spin dz2 orbital, causing a ‘turning off’ of the exchange splitting and quenching of the spin moment at the Co magnetic centers. Our investigations suggest that, by this mechanism, molecule–substrate interactions can be used to control the quenching of the magnetic moments of the adsorbed molecules.


 Please wait while we load your content...
Please wait while we load your content...