Iron assisted formation of CO2 over condensed CO and its relevance to interstellar chemistry†
Abstract
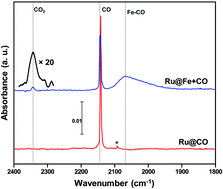
Catalytic conversion of CO to CO2 has been investigated in ultrahigh vacuum (UHV) under cryogenic conditions (10 K). This cryogenic oxidation is assisted by iron upon its co-deposition with CO, on a substrate. The study shows that the interaction of Fe and CO results in a Fe–CO complex that reacts in the presence of excess CO at cryogenic conditions leading to CO2. Here, the presence of CO on the surface is a prerequisite for the reaction to occur. Different control experiments confirm that the reaction takes place in the condensed phase and not in the gas phase. Surface sensitive reflection absorption infrared spectroscopy (RAIRS), temperature programmed desorption (TPD), and Cs+ based low energy ion scattering are utilized for this study. The iron assisted formation of CO2 may be proposed as another pathway relevant in interstellar ices, containing CO. This direct oxidation process, which occurs at extremely low temperatures and pressures, in the presence of a reactive metal species like iron (the most abundant metal in the interstellar medium) may have astrochemical importance. It does not require any external energy in the form of photo-irradiation or thermal processing. Such reactions are highly relevant in cold dense molecular clouds where interactions between neutral species are more favoured.
- This article is part of the themed collection: 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...