State-to-state photodissociation dynamics of CO2 around 108 nm: the O(1S) atom channel
Abstract
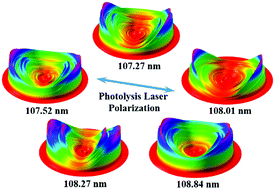
State-to-state photodissociation of carbon dioxide (CO2) via the 3p1Πu Rydberg state was investigated by the time-sliced velocity map ion imaging technique (TSVMI) using a tunable vacuum ultraviolet free electron laser (VUV FEL) source. Raw images of the O(1S) products resulting from the O(1S) + CO(X1Σ+) channel were acquired at the photolysis wavelengths between 107.37 and 108.84 nm. From the vibrational resolved O(1S) images, the product total kinetic energy releases and the vibrational state distributions of the CO(X1Σ+) co-products were obtained, respectively. It is found that vibrationally excited CO co-products populate at as high as v = 6 or 7 while peaking at v = 1 and v = 4, and most of the individual vibrational peaks present a bimodal rotational structure. Furthermore, the angular distributions at all studied photolysis wavelengths have also been determined. The associated vibrational-state specific anisotropy parameters (β) exhibit a photolysis wavelength-dependent feature, in which the β-values observed at 108.01 nm and 108.27 nm are more positive than those at 107.37 nm and 107.52 nm, while the β-values have almost isotropic behaviour at 108.84 nm. These experimental results indicate that the initially prepared CO2 molecules around 108 nm should decay to the 41A′ state via non-adiabatic coupling, and dissociate in the 41A′ state to produce O(1S) + CO(X1Σ+) products with different dissociation time scales.
- This article is part of the themed collection: PCCP Editor’s Choice, 2020

 Please wait while we load your content...
Please wait while we load your content...