Pivotal role of the redox-active tyrosine in driving the water splitting catalyzed by photosystem II†
Abstract
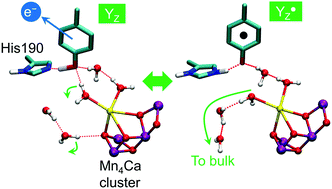
Photosynthetic water oxidation is catalyzed by the Mn4Ca cluster in photosystem II (PSII). The nearby redox-active tyrosine (YZ) serves as a direct electron acceptor of the Mn4Ca cluster and it forms a low-barrier H-bond (LBHB) with a neighboring histidine residue (D1-His190). Experimental evidence indicates that YZ oxidation triggers changes in the hydrogen bonding network that precede proton abstraction from the Mn4Ca cluster. In order to characterize such changes, we compare ab initio molecular dynamics simulations of different states of the catalytic cycle of PSII with dynamics of isolated tyrosine models (namely, p-cresol) in different oxidation states. The systematic comparison of the H-bond networks in different simulated systems suggests that the YZ oxidation leads to a water hydration pattern which is more similar to that of the neutral p-cresol rather than that of the p-cresol anion. Our simulations also reveal the twofold nature of the interactions between YZ and the Mn4Ca cluster. Firstly, the YZ oxidation triggers rapid structural changes of the H-bond pattern in the proximity of the cluster which have been observed to propagate on the ps time scale on the Ca2+ hydration shell up to other water molecules in the proximity of the cluster. Secondly, it is clear that YZ interacts with the Mn4Ca cluster also through Coulombic interactions mediated by CP43-Arg357 through the remaining positive charge of the 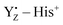 pair. Our results are able to identify, for the first time, the structural rearrangements guided by the oxidation of YZ necessary for the evolution of the water splitting reaction in PSII. Based on these findings, we propose a mechanism of structural changes which is functional towards the progression of the catalytic cycle in PSII.
pair. Our results are able to identify, for the first time, the structural rearrangements guided by the oxidation of YZ necessary for the evolution of the water splitting reaction in PSII. Based on these findings, we propose a mechanism of structural changes which is functional towards the progression of the catalytic cycle in PSII.


 Please wait while we load your content...
Please wait while we load your content...