The borderline: exploring the structural landscape of triptycene in cocrystallization with ferrocene†
Abstract
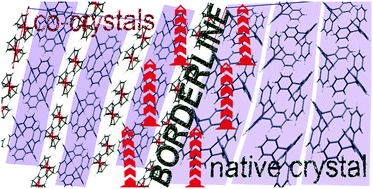
Cocrystallization of the geometrically demanding triptycene (TripH) with the compact and electron-rich ferrocene (FcH) and its derivatives afforded C–H---πCp assembled supramolecular chains of alternating (C5H5)(C5H4R)Fe and TripH molecules in a series of binary crystals: FcH/TripH (1), (C10H9)FeCHO/TripH (2), diferrocenyldiacetylene/TripH (3), paracyclophane-C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Fc/TripH (4), and (C10H9)2Fe2/TripH (5). Analysis of the structure of cocrystals 1–5 (single crystal XRD) showed significant Hbridgehead---πCp interaction along with C–H---π and π---π interactions. Upon varying the groups on FcH, the appreciable Hbridgehead---πCp interaction remains in action to maintain the efficient packing and dense arrangement in the form of properly crystallized chains among the TripH and FcH derivatives mostly. In the case of 5, the effective close packing of such chain oligomers could not be achieved, so at this point, we reached a borderline at the structural landscape of TripH. This borderline can be described as an intermediate area between the native TripH and co-crystals domains, where TripH and Fc2 molecules form 2D ribbons, which are packed closely by T-shaped C–H---πPh interactions between TripH molecules which exactly reproduce the geometry of TripH---TripH interaction in the TripH native crystal.
C–Fc/TripH (4), and (C10H9)2Fe2/TripH (5). Analysis of the structure of cocrystals 1–5 (single crystal XRD) showed significant Hbridgehead---πCp interaction along with C–H---π and π---π interactions. Upon varying the groups on FcH, the appreciable Hbridgehead---πCp interaction remains in action to maintain the efficient packing and dense arrangement in the form of properly crystallized chains among the TripH and FcH derivatives mostly. In the case of 5, the effective close packing of such chain oligomers could not be achieved, so at this point, we reached a borderline at the structural landscape of TripH. This borderline can be described as an intermediate area between the native TripH and co-crystals domains, where TripH and Fc2 molecules form 2D ribbons, which are packed closely by T-shaped C–H---πPh interactions between TripH molecules which exactly reproduce the geometry of TripH---TripH interaction in the TripH native crystal.


 Please wait while we load your content...
Please wait while we load your content...