Asymmetric Si-rhodamine scaffolds: rational design of pH-durable protease-activated NIR probes in vivo†
Abstract
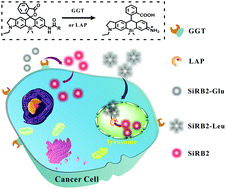
A group of asymmetric Si-rhodamine scaffolds was designed for protease-activated NIR probes. Dual pH-inertia for both spirocyclized fluorescent probes and fluorescent products of zwitterions form over a wide range of pH (4.0–11.0). Leucine aminopeptidase (LAP) and γ-glutamyl transpeptidase (GGT) were monitored by fluorescent imaging in vivo.


 Please wait while we load your content...
Please wait while we load your content...