Strategic reconstruction of macrophage-derived extracellular vesicles as a magnetic resonance imaging contrast agent†
Abstract
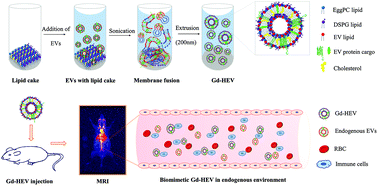
A contrast agent (CA) in magnetic resonance imaging (MRI) is now an essential add-on to obtain high-quality contrast-enhanced anatomical images for disease diagnosis and monitoring the treatment response. However, the rapid elimination of CAs by the immune system and excretion by the renal route has limited its application. As a result, the CA dose for effective contrast is ever-increasing, resulting in toxic side effects such as gadolinium (Gd) related nephrogenic systemic fibrosis (NSF) toxicity. Considering the widespread application of Gd-based CAs, it is now very important to revisit their formulation in order to improve their local concentration and minimize their dose while achieving clinical goals. Therefore, we have adapted a unique strategy to maximize Gd delivery to the target site using macrophage cell-derived extracellular vesicles (EVs) reconstructed with a Gd-conjugated liposomal system herein called gadolinium infused hybrid EVs (Gd-HEVs). We hypothesize that Gd-HEVs, owing to the presence of immune cell-derived EV protein cargo, can effectively disguise themselves as a biological entity, prolong the retention time for contrast enhancement, and show tumor specificity. Incorporation of Gd into nanoformulations can enhance the longitudinal relaxivity r1 by reducing the tumbling rate of paramagnetic metal complexes. Here, Gd-HEVs showed a higher r1 relaxivity of 9.86 mM−1 s−1 compared to 3.98 mM−1 s−1 of Magnevist® at an equivalent Gd concentration, when measured by clinical 3T MRI. This will allow us to reduce the clinically used Gd concentration about three-fold while maintaining contrast in the clinical window thereby supporting our hypothesis. Furthermore, Gd-HEVs showed a preferential cellular interaction and accumulation towards cancer cells compared to non-cancer cells, both in vitro and in vivo. More importantly, Gd-HEVs showed excellent contrast enhancement in the blood vasculature with a higher retention time compared to its counterpart, Magnevist®. Our study successfully showed that the incorporation of Gd in the EV framework can help to enhance the contrast ability, and therefore it can be a platform technology for the development of safer MRI contrast agents.


 Please wait while we load your content...
Please wait while we load your content...