Generation of TD50 values for carcinogenicity study data†
Abstract
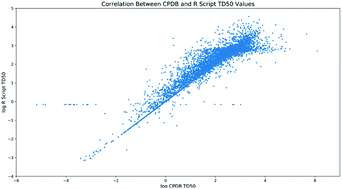
Carcinogenic potency is a key factor in the understanding of chemical risk assessment. Measures of carcinogenic potency, for example TD50, are instrumental in the determination of metrics such as the threshold of toxicological concern (TTC), acceptable intake (AI) and permitted daily exposure (PDE), which in turn impact on human exposure. The Carcinogenic Potency Data Base (CPDB) has provided a source of study information, complete with calculated TD50 values. However, this is no longer actively updated. An understanding of carcinogenic potency, which can be derived from dose–response data, can be used as part of human risk assessments to generate safety thresholds under which cancer risk is judged to be minimal. The aim of this paper is to produce a transparent methodology for calculating TD50 values from experimental data in a manner consistent with the CPDB. This was then applied across the same data as used in the CPDB and analysis done on the correlation with the CPDB TD50 values. While the two sets of values showed a high level of correlation overall, there were some significant discrepancies. These were predominantly due to a lack of clarity in the CPDB methodology and inappropriate use of a linear model in TD50 calculation where the data was not suitable for such an approach.


 Please wait while we load your content...
Please wait while we load your content...