Photoluminescent manipulation of phenoxazine-based molecules via regulating conformational isomerization, and the corresponding electroluminescent properties†
Abstract
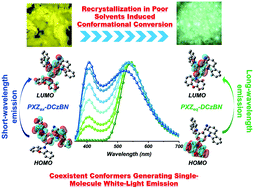
Multi-stimuli-responsive materials have attracted tremendous attention owing to their promising applications. Herein, we constructed two multifunctional optoelectronic materials (PXZ-DCzBN and PXZ-DtCzBN) composed of benzonitrile, phenoxazine and carbazole or tert-butyl carbazole moieties, which are endowed with thermally activated delayed fluorescence and mechanochromic luminescence characteristics. Intriguingly, under external stimuli, PXZ-DCzBN presents a unique multicolor transformation, including single-molecule white-light emission, while PXZ-DtCzBN simply displays bicolor switching. The white-light emission of PXZ-DCzBN is eventually attributed to the coexistence of two different conformers: ‘quasi-equatorial’ (eq) and ‘quasi-axial’ (ax). PXZeq-DCzBN can convert into PXZax-DCzBNvia recrystallization in poor solvents. PXZeq-DCzBN generates long-wavelength emission from an intramolecular charge transfer excited state, and PXZax-DCzBN emits short-wavelength fluorescence through a locally excited transition. This study evaluates the importance of the conformational isomerization of PXZ moieties and provides an innovative perspective for understanding the relationship between molecular conformations and excited-state characteristics.


 Please wait while we load your content...
Please wait while we load your content...