Donor–acceptor copolymers with 1,7-regioisomers of N,N′-dialkylperylene-3,4,9,10-tetracarboxydiimide as materials for photonics†
Abstract
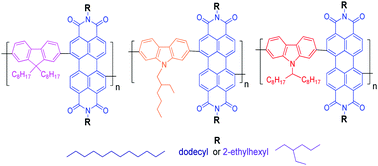
Donor–acceptor (D–A) copolymers containing N,N′-dialkylperylene-3,4,9,10-tetracarboxydiimide electron-acceptor units with dodecyl (DDPDI) or 2-ethylhexyl (EHPDI) substituents and three different electron-donor units (9,9-dioctylfluorene, 9-(2-ethylhexyl)carbazole or 9-(heptadecan-9-yl)carbazole) were synthesized by Suzuki coupling and were then characterized. Their thermal, photophysical, electrochemical and spectroelectrochemical properties were studied. The effects of the alkyl side chain combination on the properties were evaluated and are discussed. Despite large differences in the absorption spectra of the individual DDPDI and EHPDI units or monomers in solutions and thin films, the absorption spectra of the copolymers in solutions and thin films exhibited similar shapes. More pronounced side chain effects were observed in the photoluminescent (PL) behaviour than in the absorption behaviour. Different alkyl side chains on the D–A copolymers similarly affected the visible absorption band contributions in the solutions and thin films, but the exhibited effects were different for the PL spectra and efficiency. The copolymers exhibited reversible reduction and irreversible oxidation. The ionization potential and bandgap values were influenced by the donor unit and by the perylene-3,4,9,10-tetracarboxydiimide (PDI) side chain nature. Spectroelectrochemical changes are reported, and potential sensing applications are outlined.


 Please wait while we load your content...
Please wait while we load your content...