Organic solvent-assisted synthesis of the K3SiF7:Mn4+ red phosphor with improved morphology and stability†
Abstract
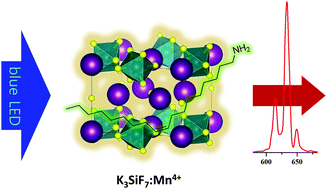
Recently, a novel host, K3SiF7, for Mn4+-activated phosphors was discovered and it was revealed that Mn4+-doped K3SiF7 has an advantage in decay time compared with the most widely used Mn4+-doped K2SiF6. Here, we developed an organic solvent-assisted synthetic method for the K3SiF7:Mn4+ phosphor. To the best of our knowledge, it is the first publication about a newly developed synthetic method of the K3SiF7:Mn4+ phosphor. Various organic solvents with different functional groups have been tried to synthesize the K3SiF7:Mn4+ phosphor. When we introduced oleylamine (OAm) as an organic solvent, uniform K3SiF7:Mn4+ particles below 5 μm were obtained at a lower temperature than the conventional solid state reaction (SSR). As a result of thermal quenching (TQ) and 85/85 tests, it was confirmed that despite its small size, the resulting K3SiF7:Mn4+ has enhanced stability against heat and moisture with the aid of an organic protecting layer. We expect that this organic solvent-assisted synthesis can be applied to the synthesis of other phosphors with a chemical formula of A3MF7:Mn4+ (A = alkali metal ion; M = Si, Ge, Zr, etc.).


 Please wait while we load your content...
Please wait while we load your content...