Predicting the yield of ion pair formation in molecular electrical doping: redox-potentials versus ionization energy/electron affinity†
Abstract
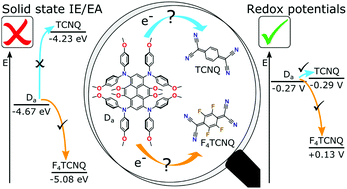
Efficient electrical doping of organic semiconductors relies on identifying appropriate molecular dopants that are capable of ionizing semiconductor molecules with a high yield, thereby creating mobile charges. We explore the suitability of two different material parameters to predict ion pair formation for different sets of semiconductor–dopant combinations: (i) redox-potentials measured by cyclic voltammetry in solution and (ii) ionization energy (IE)/electron affinity (EA) measured on thin films by ultraviolet/inverse photoelectron spectroscopy. Our study suggests, at least for molecular semiconductors and dopants, that redox-potentials are better suited to identify matching material pairs and their ion pair formation yield than IE/EA values. This is ascribed to the dependence of IE/EA values on molecular orientation and film structure on and above the meso-scale. In contrast, cyclic voltammetry measurements, although performed on solutions rather than on thin films, capture dopant–semiconductor energy levels on the molecular scale, which is more relevant for doping even in the case of solid thin films.


 Please wait while we load your content...
Please wait while we load your content...
